Subtotal ¥0.00
FDA regulations for medical wristbands explained Class I status 510k exemptions UDI labeling GMP ISO material safety and LinkWin compliance
The Legal Foundation: FDA Classification
Navigating the maze of FDA Regulations for Medical Wristbands can feel overwhelming, but it starts with a simple question: Is a wristband actually a medical device? The short answer is yes. If you are manufacturing, importing, or distributing these products for US hospitals, you aren’t just selling office supplies; you are dealing with regulated medical equipment. Understanding where we stand legally is the first step to ensuring we don’t face costly recalls or import detentions.
Defining the Device: 21 CFR 880.6025
The FDA doesn’t leave room for guessing. Under the regulation 21 CFR 880.6025, a patient identification wristband is explicitly defined as a device intended to be worn by a patient to identify them.
This specific code is critical because it categorizes the product under “General Hospital and Personal Use Devices.” It confirms that even a simple thermal band is subject to federal oversight. When we look at compliance, we always start by referencing this code to ensure we are speaking the same language as the regulators.
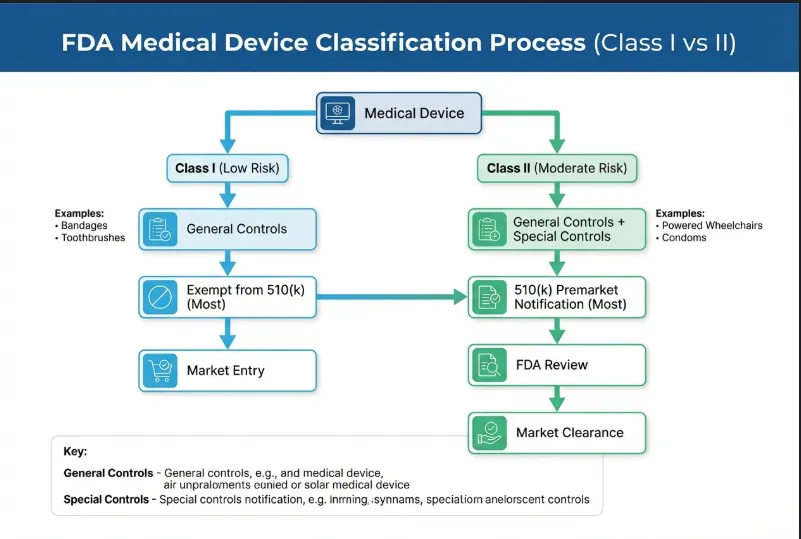
Class I vs. Class II Distinction
Not all medical devices are created equal. The FDA categorizes them based on risk, and understanding this distinction saves a lot of headaches.
- Class I Medical Devices: This is where most standard patient ID wristbands fall. They are considered low risk to the patient. The controls required are general, focusing on basic safety and accurate labeling.
- Class II Medical Devices: These carry a moderate risk and require tighter controls. While rare for standard ID bands, if a wristband includes active monitoring sensors or specialized software, it could be bumped up to this category.
For the vast majority of the products we deal with, we operate strictly within the Class I framework. This means the regulatory barrier to entry is lower, but the responsibility for quality remains high.
Understanding the 510(k) Exemption
Here is the best news for manufacturers and suppliers in this niche: most standard medical wristbands benefit from a Class I Medical Device Exemption.
This means they are exempt from the 510(k) Premarket Notification requirement. In plain English, you generally do not need to submit a massive dossier of paperwork to the FDA proving your device is “substantially equivalent” to a predicate device before you can market it.
However, there are caveats to this exemption:
- Intended Use: The device must only be used for identification.
- Technology: If you introduce fundamental changes in technology (like novel materials that haven’t been tested for skin contact), the exemption might be voided.
We leverage this exemption to speed up time-to-market, but we never mistake “exempt” for “unregulated.” We still have to follow the general controls to keep that status valid.
General Controls: What Compliance Actually Looks Like
Even though most patient ID bands are exempt from the rigorous 510(k) premarket notification, that doesn’t mean the industry is unregulated. We still have to strictly follow “General Controls.” These are the baseline requirements set by the government to ensure that FDA Regulations for Medical Wristbands are actually effective in the real world. Being exempt isn’t a free pass; it just means the focus shifts to how we run our business and manufacturing lines daily.
Establishment Registration & Listing (Product Code FNM)
The first step in compliance is simply raising our hand and telling the FDA we exist. You cannot legally market a medical device in the US without completing FDA Establishment Registration. This applies to both domestic manufacturers and foreign exporters.
When we register, we have to list every specific device we make. For patient identification bands, we use the FDA Product Code FNM. This code categorizes the product specifically as a “Wristband, Patient Identification.” It ensures that when regulators look at our inventory, they know exactly what safety standards apply to our products.
Good Manufacturing Practices (GMP) & QSR
Quality isn’t just about the final product looking good; it’s about the process being consistent. We adhere to Good Manufacturing Practices (GMP), which are outlined in 21 CFR Part 820 Quality Systems. This regulation requires us to have a quality management system (QMS) in place.
We don’t just guess during production. We follow strict protocols to ensure:
- Consistency: Every wristband in a batch meets the same durability and safety specs.
- Environment Control: Manufacturing areas are clean and suitable for producing medical-grade items.
- Management Responsibility: Our leadership is directly involved in reviewing quality audits.
Record Keeping & Complaint Handling
If something goes wrong, we need to know why, how, and when. General controls mandate rigorous record-keeping. We track the history of our devices from the raw material stage all the way to distribution. This traceability is crucial if a recall ever becomes necessary.
Furthermore, we take feedback seriously. If a hospital reports that a band caused skin irritation or fell off, we document it. Under specific circumstances involving adverse events, we are required to follow Medical Device Reporting (MDR) procedures. This system ensures that any potential risks to patient safety are flagged, investigated, and resolved immediately rather than being swept under the rug.
Material Safety and Biocompatibility Standards
When we navigate FDA Regulations for Medical Wristbands, the focus isn’t just on the data printed on the band, but on the material itself. Because these devices are in prolonged contact with patient skin, the FDA requires us to prove that the materials are biologically safe. We don’t just pick a durable plastic; we select medical-grade materials that won’t harm the patient.
ISO 10993 Standards for Skin Contact
The industry benchmark for this is ISO 10993 Biocompatibility. Compliance with this standard is critical for any device touching intact skin. It ensures that the wristband is chemically stable and biologically inert. We focus on three primary tests to meet these requirements:
- Cytotoxicity: Verifying that the material does not damage or kill cells.
- Sensitization: Ensuring the material doesn’t trigger an allergic immune response after repeated exposure.
- Irritation: Confirming that the band won’t cause redness, swelling, or rashes upon contact.
Chemical Safety and Latex-Free Mandates
In a hospital environment, minimizing allergens is non-negotiable. This is why Latex-free Hospital Wristbands have become the standard. Under FDA labeling guidelines, if we claim a product is latex-free, we must ensure the entire supply chain is free from natural rubber latex proteins. This prevents life-threatening anaphylactic reactions in sensitized patients. We also monitor for other hazardous chemicals, ensuring compliance with global safety standards regarding phthalates and BPA.
Antimicrobial Coatings and Claims
Adding antimicrobial additives to wristbands is a common request, but FDA regulation draws a hard line on how these are marketed.
- Product Protection: If the coating is intended solely to protect the wristband itself from odors, staining, or deterioration caused by bacteria, it is generally regulated as a treated article.
- Health Claims: If a manufacturer claims the wristband actively “kills bacteria to prevent patient infection,” the FDA treats this as a medical claim. This requires significantly more data and regulatory scrutiny.
We stick to strict definitions to ensure that any antimicrobial feature provides value without overstepping regulatory boundaries or misleading healthcare providers.
Labeling and Unique Device Identification (UDI) Rules
When we talk about FDA Regulations for Medical Wristbands, accurate labeling is just as critical as the material itself. It is not enough to simply manufacture a safe product; the information accompanying it must meet strict federal standards to ensure traceability and proper usage.
21 CFR Part 801 Labeling Requirements
Under 21 CFR Part 801, general labeling provisions require us to be completely transparent about the device’s origin and function. We treat Medical Device Labeling Requirements seriously to prevent any confusion in a hospital setting. The FDA mandates that our packaging is legible and includes specific data points.
Key Labeling Elements We Include:
- Manufacturer Information: The name and place of business of the manufacturer, packer, or distributor.
- Intended Use: A clear statement describing what the device is for (e.g., patient identification).
- Adequate Directions: Instructions ensuring the device is used safely and effectively.
- English Language: All required information must appear in English for the US market.
Do Wristbands Need a UDI Barcode?
This is one of the most common questions we get regarding FDA Regulations for Medical Wristbands. The short answer is yes, but the implementation depends on the device class. Most medical wristbands fall under Class I.
The Unique Device Identification (UDI) system was established to adequately identify medical devices through their distribution and use. However, for single-use patient ID bands, the requirements often focus on the packaging rather than the individual band itself.
UDI Implementation Breakdown:
| Requirement Level | Description |
|---|---|
| Device Identifier (DI) | A fixed portion that identifies the labeler and the specific version or model of the device. |
| Production Identifier (PI) | A variable portion that identifies the lot or batch number, expiration date, and manufacturing date. |
| Direct Marking | Exempt. Most single-use wristbands do not require a UDI directly on the band itself, provided the UDI is on the packaging (box/shelf pack). |
| GUDID Submission | We must submit data concerning the device to the FDA’s Global Unique Device Identification Database (GUDID). |
By adhering to these UDI standards, we ensure that every box of wristbands can be traced back to its manufacturing batch, significantly improving safety management and recall efficiency if necessary.
Beyond the FDA: The Joint Commission (TJC) & Usage
Compliance doesn’t stop once the product leaves the manufacturing plant. While I focus heavily on meeting FDA Regulations for Medical Wristbands, the actual application in hospitals is strictly monitored by The Joint Commission (TJC). You can have a fully registered Class I device, but if it doesn’t perform reliably in a clinical setting, it fails the end-user.
National Patient Safety Goals (NPSG.01.01.01)
The gold standard for hospital accreditation is adhering to Joint Commission NPSG.01.01.01. This specific goal focuses entirely on improving the accuracy of patient identification. The rule is simple: healthcare providers must use at least two patient identifiers (such as name and date of birth) when providing care, treatment, or services.
For us, this means the wristband is the critical link. It acts as the primary source of truth. If a nurse cannot verify these details because of a poor-quality band, the hospital faces immediate citation for Patient Identification Errors. The wristband must support the hospital’s workflow, ensuring that the “two identifiers” are always attached to the patient.
Physical Quality and Patient Safety
Regulatory compliance in a hospital environment relies heavily on the physical durability of the ID band. A wristband that falls off or becomes illegible is a safety hazard. We prioritize Thermal Wristband Durability because these bands face harsh conditions, from constant friction against bedsheets to frequent exposure to alcohol-based hand sanitizers.
To ensure patient safety and TJC compliance, the wristband must deliver:
- Scannability: Barcodes must remain crisp and readable for medication administration (BCMA) systems.
- Resistance: The print cannot fade or smear when exposed to water, soap, or blood.
- Security: The closure must be tamper-evident so it cannot be transferred between patients.
If the data on the wristband is compromised, the patient’s safety is compromised. That is why physical quality is just as much a regulatory requirement as the paperwork.
Ensuring Compliance with a Trusted Partner
Navigating FDA Regulations for Medical Wristbands is not a solo journey. You need a supplier who understands that a wristband is a medical device, not just a piece of paper. We treat compliance as the baseline of our production, ensuring that your facility remains audit-ready and patient-safe.
Quality Assurance and GMP Alignment
We don’t leave quality to chance. Our production lines operate under strict Good Manufacturing Practices (GMP) to meet 21 CFR Part 820 Quality Systems requirements. This means every step, from raw material sourcing to final cutting, is documented and controlled.
When you partner with us, you get:
- Traceability: Every batch is tracked to ensure accountability.
- Consistency: Strict adherence to FDA Establishment Registration standards means the product quality never fluctuates.
- Audit Readiness: Our records support your compliance needs during inspections.
Material Excellence for Hospital Environments
A compliant wristband must survive the harsh reality of a hospital environment. We engineer our products for Thermal Wristband Durability, ensuring that patient data remains legible despite exposure to water, sanitizers, and friction.
Our material standards include:
- ISO 10993 Biocompatibility: Certified safe for prolonged skin contact.
- Chemical Resistance: Withstands alcohol and soaps without fading.
- Latex-free Hospital Wristbands: Eliminates the risk of allergic reactions for sensitive patients.
Customization for HIS Integration
Compliance extends to how well the device functions within your ecosystem. We customize our wristbands to integrate seamlessly with major Hospital Information Systems (HIS) like Epic, Cerner, and Meditech.
Precise manufacturing ensures that your printers produce sharp, scannable barcodes every time. This high-quality output is critical for maintaining accurate Unique Device Identification (UDI) protocols and preventing Patient Identification Errors at the point of care.
Frequently Asked Questions About Wristband Regulations
Navigating the landscape of FDA Regulations for Medical Wristbands can raise a lot of specific questions. Here are the answers to the most common inquiries we receive from procurement officers and distributors.
Do Medical Wristbands Have an Expiration Date?
Technically, the wristband material itself—usually durable synthetic or plastic—does not expire like medication. However, performance factors do have a shelf life.
- Adhesives: The glue on the closure can dry out over long periods, especially if stored in hot warehouses.
- Thermal Coating: For direct thermal bands, the chemical coating required for printing can degrade, affecting Thermal Wristband Durability.
We generally recommend using stock within two years to ensure the print remains crisp and the adhesive secures the patient ID properly. Always check the manufacturer’s recommended storage conditions.
Can I Import Wristbands Without FDA Registration?
No. If the wristbands are intended for medical purposes (patient identification in hospitals), you cannot skip this step. Even though they fall under the Class I Medical Device Exemption, the manufacturer and the initial importer must have an active FDA Establishment Registration.
Customs officials frequently detain shipments that lack a valid registration number. Attempting to bypass this requirement puts your supply chain at risk and violates federal law.
What is the FDA Product Code for Wristbands?
Correct classification is vital for compliance. For standard patient identification bands, the specific identifier is:
- Product Code: FNM (Holder, Wrist, Patient)
- Regulation Number: 21 CFR 880.6025
Using the FDA Product Code FNM ensures your device listing is accurate and signals to regulators that the product is a Class I exempt device, simplifying the import and sales process.
Related Sources
https://www.fda.gov/medical-devices/overview-device-regulation/general-controls-medical-devices
https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?CFRPart=801

